Every morning, millions of Americans add a scoop of green powder to their smoothie, their water, or their yogurt. The label says things like "superfood," "nutrient-dense," and "plant-based wellness." One of the most popular ingredients driving that ritual is moringa, a plant that has been used in traditional medicine for centuries and has more recently become a fixture in health food stores and wellness apps.
In early 2026, that trusted morning routine made nearly 100 people sick across the United States. Some were hospitalized. And in one of the two simultaneous outbreaks tied to moringa products, doctors faced a situation they had never encountered before with a food-related illness: a strain of Salmonella that was resistant to every single antibiotic available to treat it.
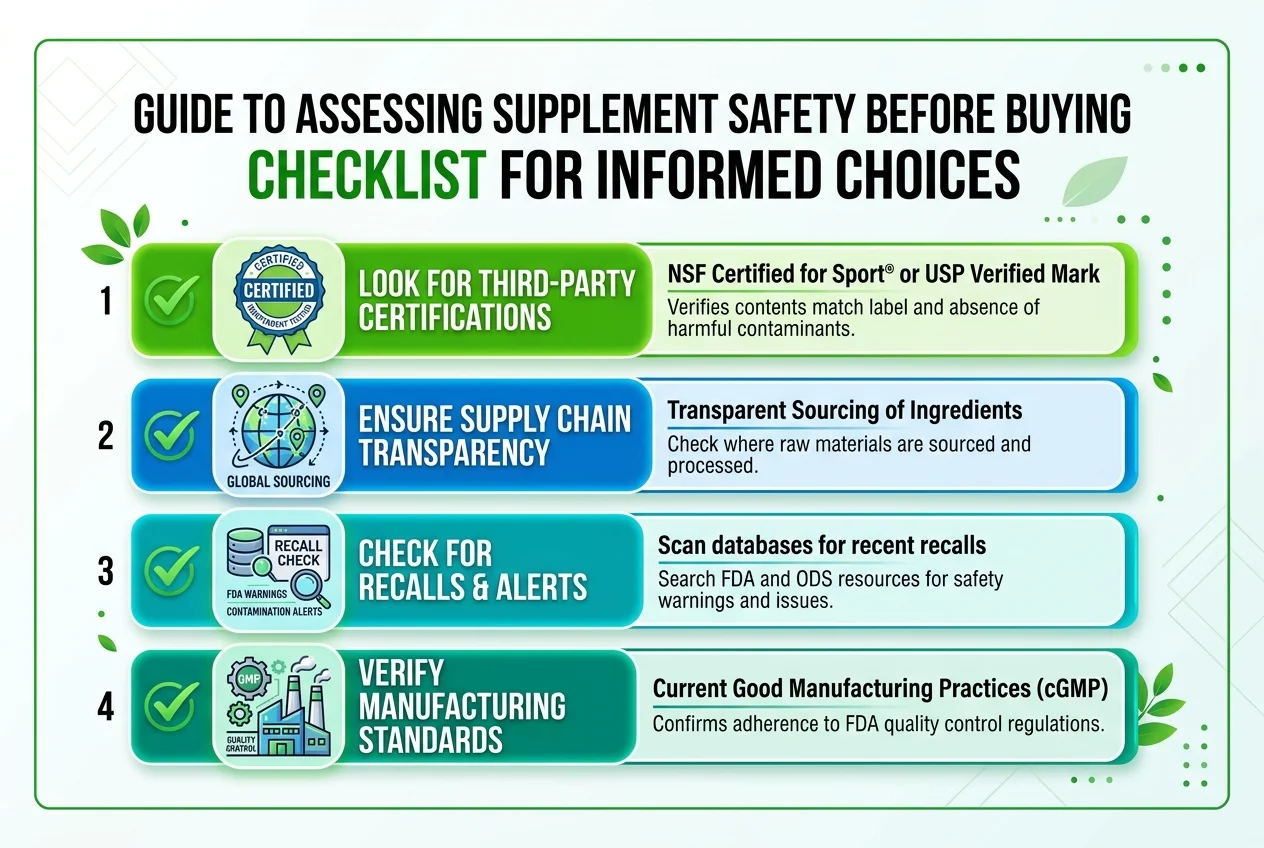
This is not a story about a dangerous chemical additive or a rogue manufacturer caught cutting corners. It is a story about a genuinely popular, genuinely nutritious ingredient and the structural vulnerabilities of the supplement supply chain that allowed contaminated products to reach consumers in 32 states.