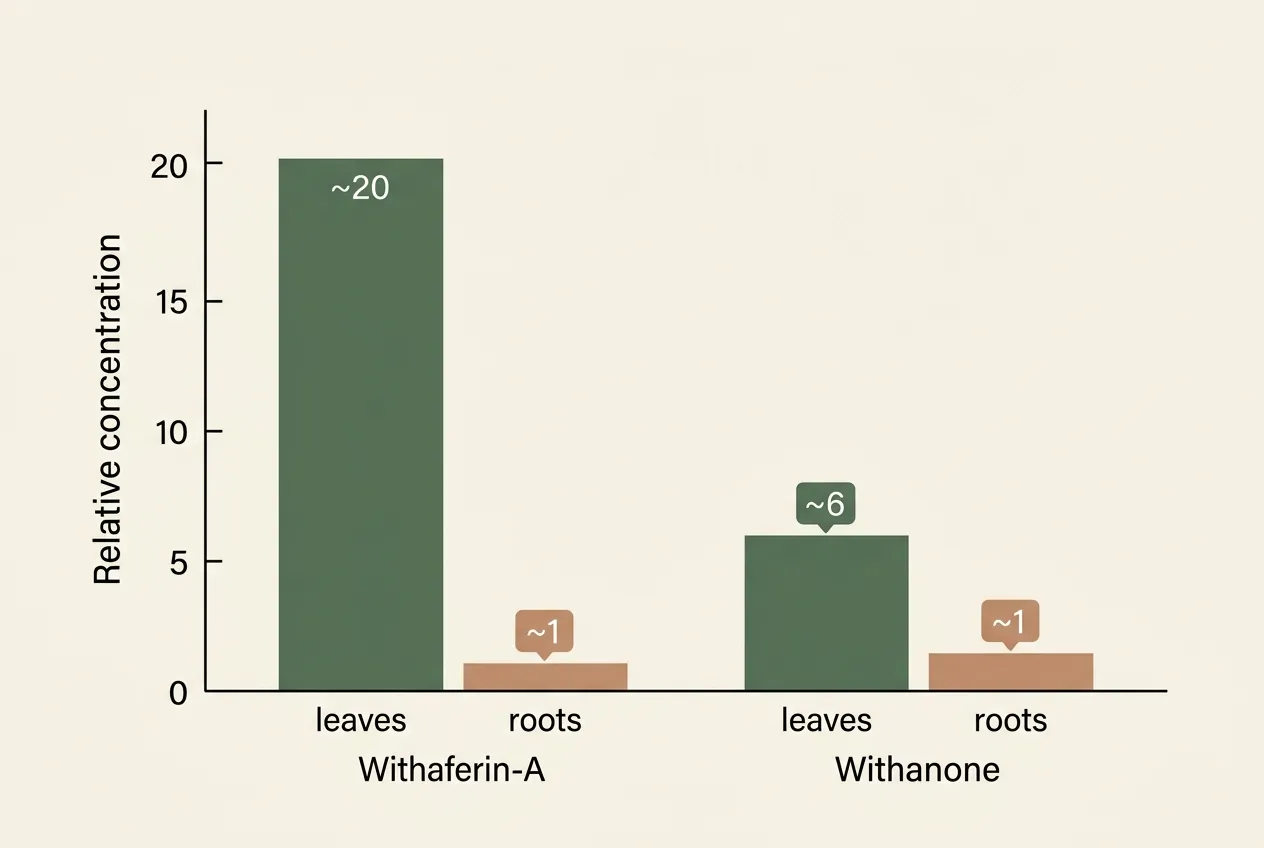
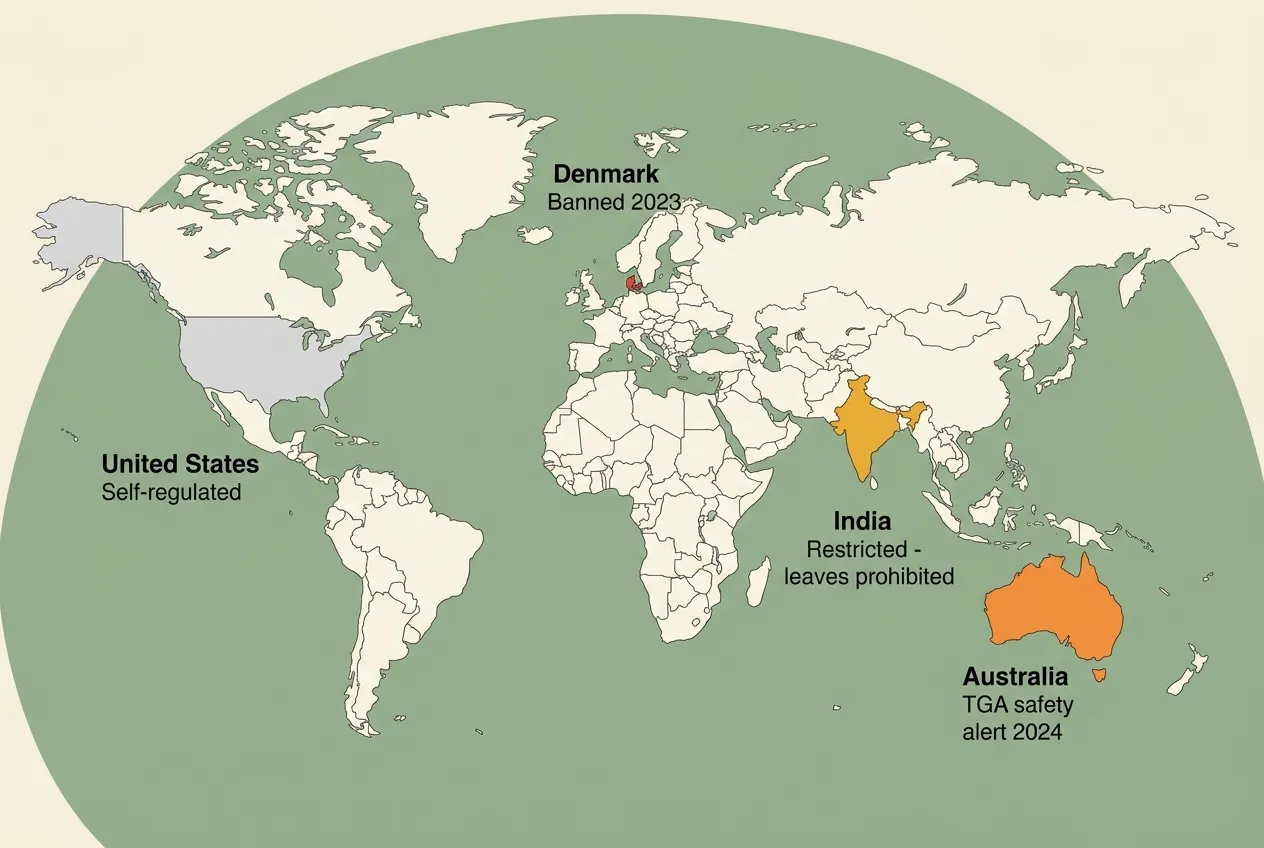
On April 16, 2026, India's food regulator quietly closed a loophole that had let parts of a booming wellness market run ahead of the rulebook. The Food Safety and Standards Authority of India (FSSAI) advisory told food business operators to stop using ashwagandha leaves in any form, crude, extract, or powder, in food products sold in India. Only the root and its extract are allowed, and only under the strict conditions already spelled out in Schedule IV of the 2016 Nutraceutical Regulations.
This matters far beyond India. Ashwagandha has become one of the most widely sold adaptogens on the planet, showing up in protein powders, stress-relief gummies, sleep drinks, and "functional" coffees. If you have ever picked up a bottle that simply says "Ashwagandha Extract" on the front, this advisory is a good reason to read the back more carefully.