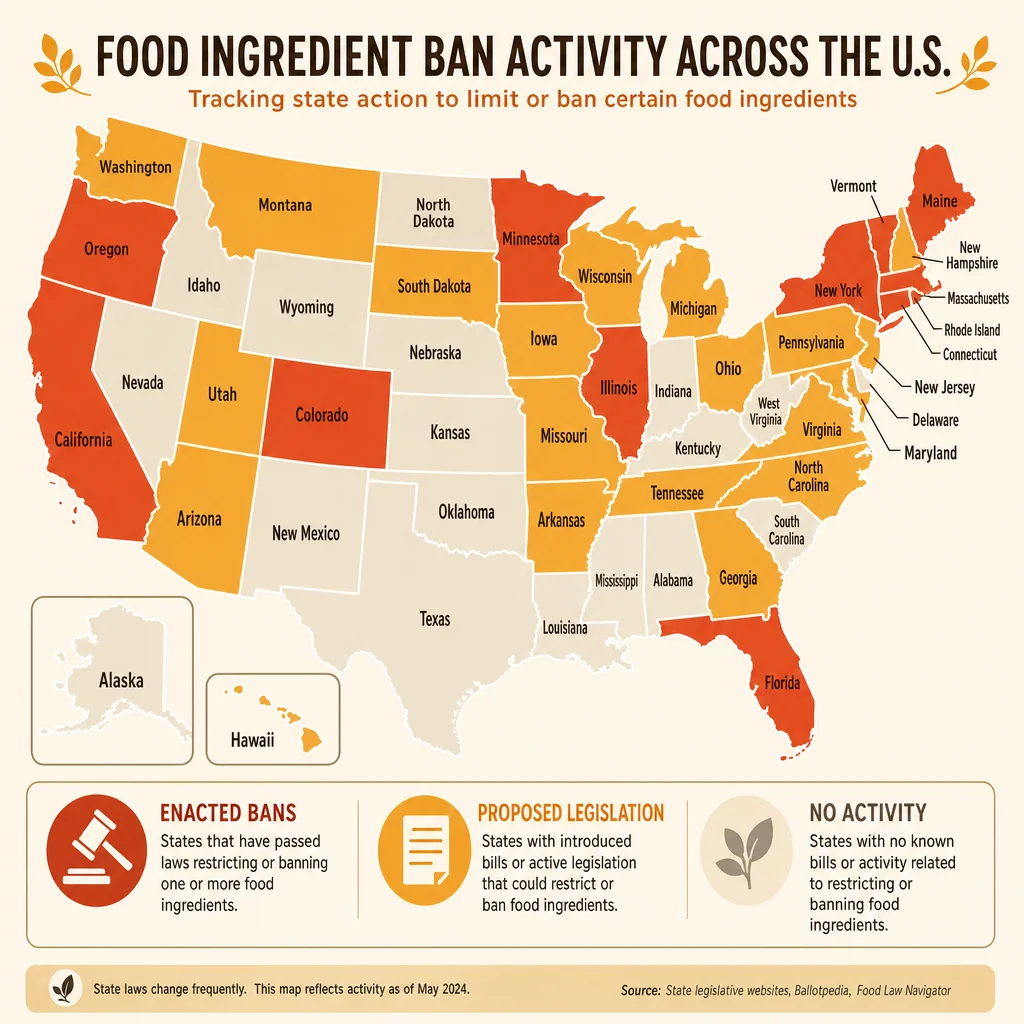
The FRESH Act amends the Federal Food, Drug, and Cosmetic Act in three major ways.
Mandatory GRAS Notification
Currently, companies can voluntarily submit a GRAS notification to the FDA. The FRESH Act would make notification mandatory. Manufacturers would be prohibited from introducing any food containing a new GRAS substance unless a notification is on file with the FDA.
The bill requires manufacturers to submit detailed information including the substance's identity, manufacturing process, specifications, and a scientific summary supporting its safety. The FDA would have 90 days to review each submission. If the agency does not respond within that window, the notification becomes effective automatically.
Critics see the 90-day provision as a critical weakness. Melanie Benesh, EWG's Vice President for Government Affairs, warned that it would "allow new chemicals to be added to food if the FDA does not respond to a GRAS notice within 90 days." The concern is that an underfunded FDA may not have the resources to review submissions thoroughly within the timeframe.
A Public GRAS Registry
The FRESH Act would create a public registry of all GRAS determinations. Within 120 days of a GRAS notification taking effect, the FDA would be required to publish the full text of the submission, excluding properly claimed confidential business information. Crucially, the bill specifies that "pivotal safety data" cannot be claimed as confidential.
For consumers, this means greater visibility into what chemicals are in their food and what safety data exists to support their use. Currently, there is no single public resource where you can look up whether a food ingredient has been reviewed for safety.
A New Category: "Common Food Ingredients"
One of the more controversial provisions creates a new statutory category called "common food ingredients." These are defined as ingredients that were "ordinarily consumed as food or available to purchase as an independent article of food on or before January 1, 1958." The list explicitly includes fruits, vegetables, legumes, nuts, seeds, algae, meat, poultry, fish, grain ingredients, milk, honey, and traditional bacterial cultures, along with any substances derived from them.
These ingredients would be excluded from regulation as food additives, effectively creating a broad categorical exemption from premarket clearance. While the intention may be to prevent common foods from being regulated like synthetic chemicals, critics worry the "derived from" language could create new loopholes.