The GRAS designation creates a widespread misconception: that if a substance is labeled GRAS, the FDA has determined it to be safe. This is not the case. GRAS means that a company, not the government, has concluded that its substance is safe for its intended use, based on evidence that the company itself compiled.
Several important caveats often get lost:
GRAS is not the same as FDA approval. A GRAS determination does not involve the pre-market review, safety testing requirements, or formal approval process that actual food additives must go through.
GRAS does not mean safe at any level. A substance may be GRAS at one concentration but potentially harmful at another. The GRAS determination is specific to conditions of intended use, but once a substance enters the food supply, there is little oversight of how widely or heavily it is actually used.
Safety evidence can be incomplete. Even for substances that have gone through the voluntary notification process, the FDA acknowledges that "safety judgments were made in the absence of a complete safety study dataset." Some GRAS determinations rely on studies conducted decades ago, using methods that would not meet today's scientific standards.
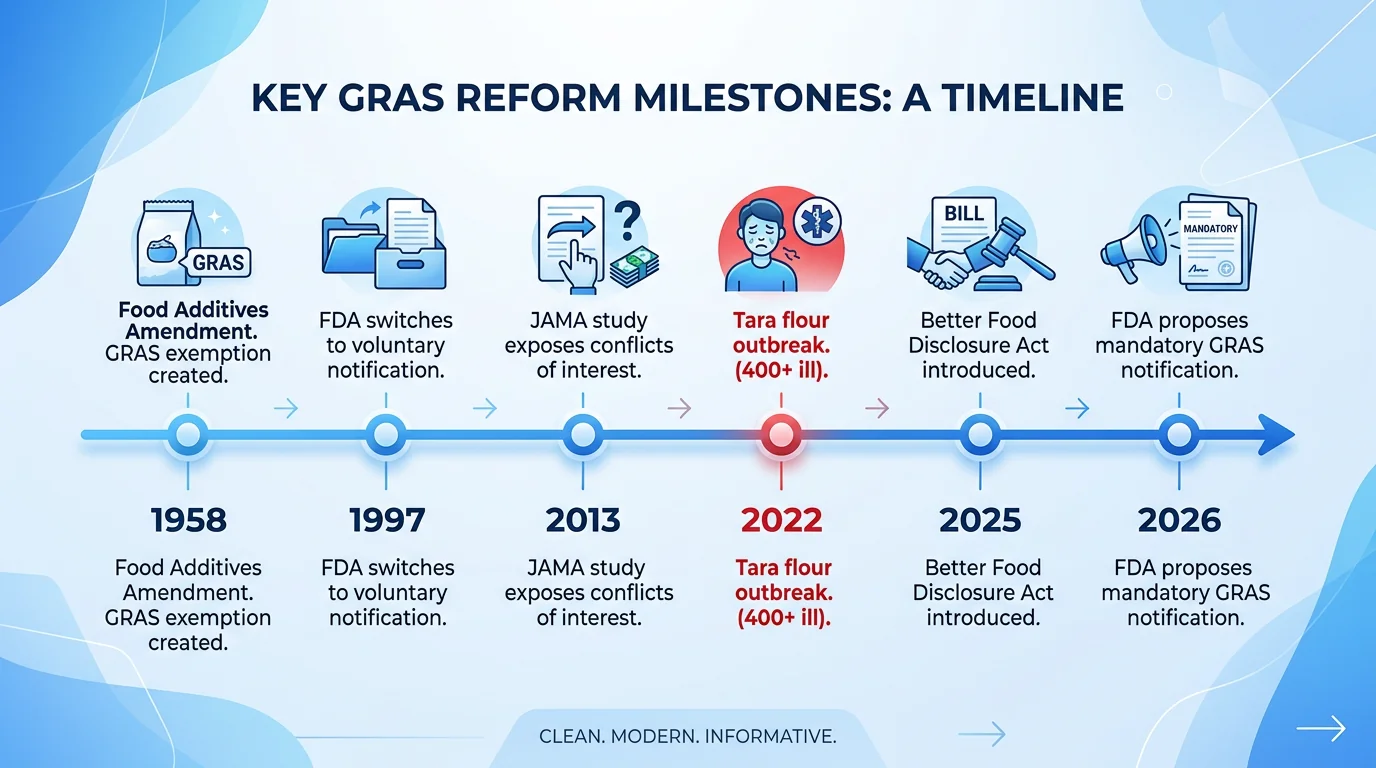
Conflicts of interest are systemic. The experts who evaluate GRAS safety evidence are often selected and paid by the company seeking the determination. A 2013 study published in JAMA Internal Medicine found that in 100% of the GRAS determinations examined, every member of the expert panel had a financial relationship with the company making the determination. Zero percent of panels included an independent member with no industry ties.