The concern about titanium dioxide in food is not about toxicity in the conventional sense. Eating a single piece of candy with TiO2 does not cause acute harm. The concern is more subtle: the possibility that long-term, chronic exposure at even low levels may cause DNA damage, and that some of the particles involved are nanoscale.
Genotoxicity: The Key Concern
Modern food-grade titanium dioxide is a mixture of particle sizes, a significant portion of which fall into the nanoscale range (below 100 nanometers). Nanoparticles behave differently from larger particles: they can penetrate cells, cross biological barriers, and interact with cellular machinery in ways that larger particles cannot.
The European Food Safety Authority's 2021 updated safety assessment, the most comprehensive review to date, concluded:
"Taking into account all available scientific studies and data, the Panel concluded that titanium dioxide can no longer be considered safe as a food additive. A concern for genotoxicity cannot be ruled out."
EFSA did not conclude that E171 is definitively harmful or that it causes cancer. What it concluded was that it could not rule out the possibility that TiO2 damages DNA or chromosomes. Under EU food safety law, uncertainty about genotoxicity is sufficient grounds for removal from the market. If regulators cannot establish a safe intake level (genotoxic effects theoretically have no safe threshold), authorization cannot be maintained.
The International Agency for Research on Cancer (IARC) has classified titanium dioxide as Group 2B — possibly carcinogenic to humans, primarily based on inhalation studies in occupational settings. The same Group 2B classification applies to potassium bromate, another additive banned in the EU but still permitted in US food. The food ingestion route involves different exposure dynamics, but the IARC classification reflects the broader scientific caution around the compound.
Gut Health and the Microbiome
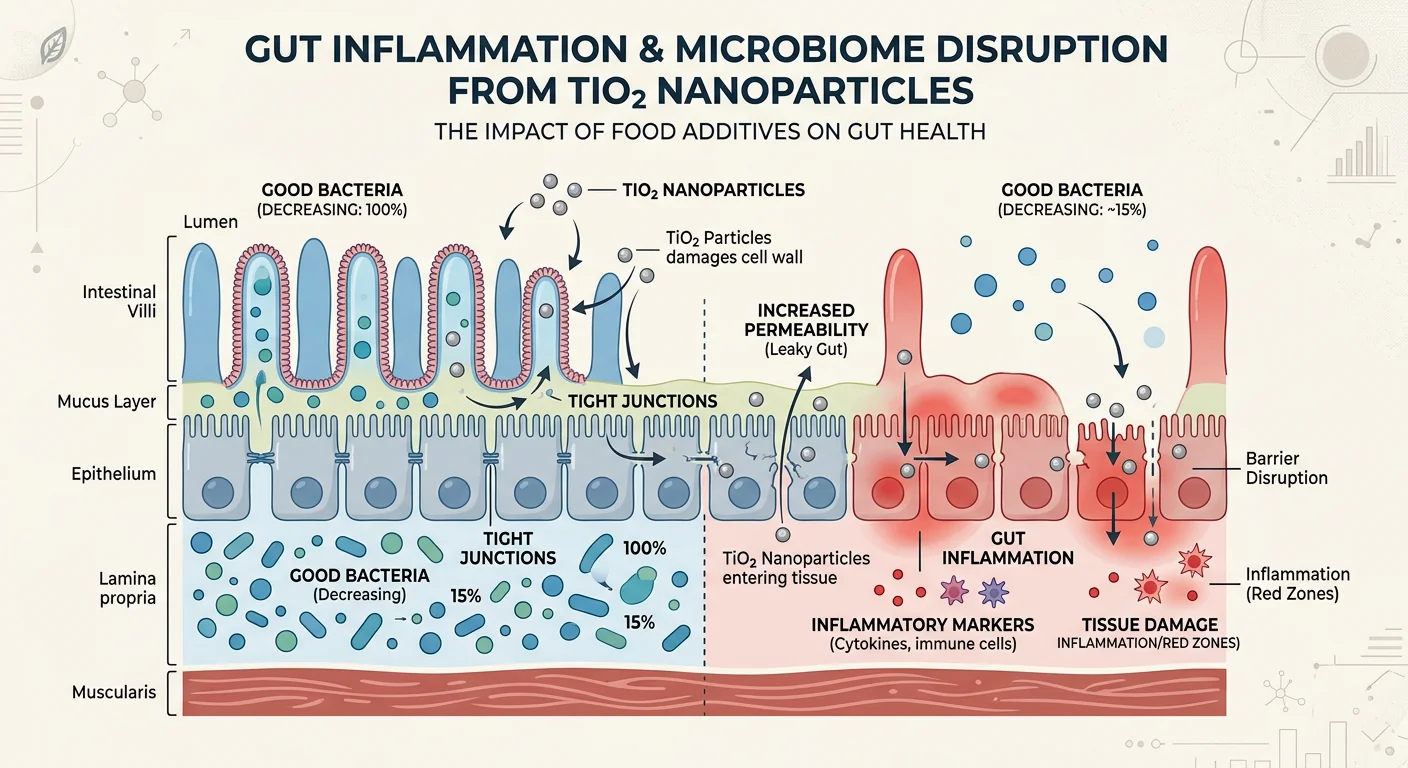
A separate body of research has examined what happens to the gut when titanium dioxide nanoparticles pass through the digestive system. Rodent studies have consistently shown:
- Gut microbiota disruption: TiO2 nanoparticles alter the composition of intestinal bacteria, reducing beneficial species such as Bifidobacterium and Lactobacillus while increasing pro-inflammatory bacterial populations
- Intestinal inflammation: Multiple studies found increased levels of inflammatory markers in the colonic mucosa following dietary TiO2 exposure
- Reduction in short-chain fatty acids (SCFAs): SCFAs are key metabolites produced by gut bacteria that support colon health and immune regulation; TiO2 exposure has been associated with their decrease
- Intestinal barrier disruption: Studies found reduced mucus production and damage to the cells lining the gut wall
A 2021 systematic review of animal studies published in Nutrients concluded that TiO2 at food-relevant doses "may promote intestinal inflammation, alter microbiota composition and metabolic activity, and affect gut barrier integrity."
The caveat is that most of this research has been conducted in rodents, and the particle sizes and doses used do not always match real-world human food exposure. Human studies remain limited. This scientific uncertainty is precisely why regulatory bodies disagree on what to do about it.