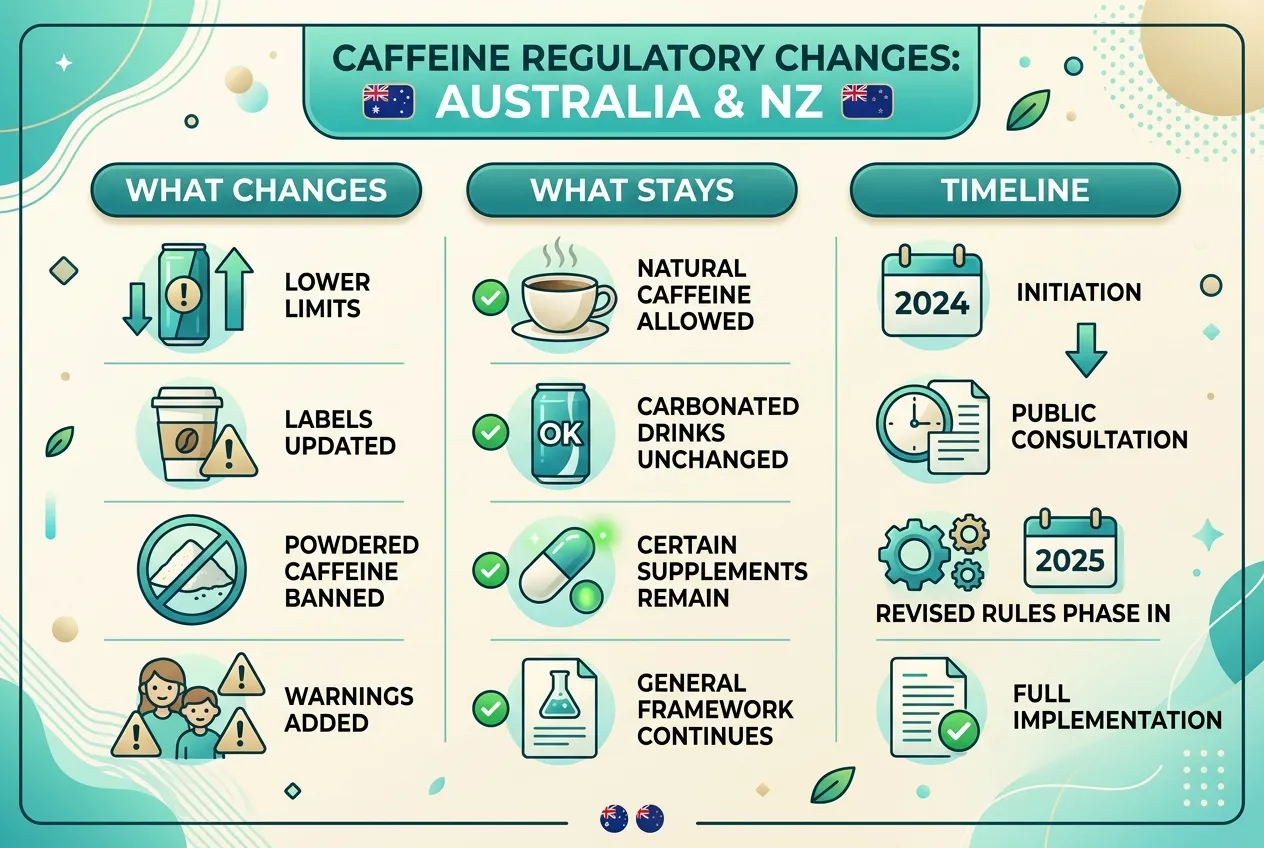
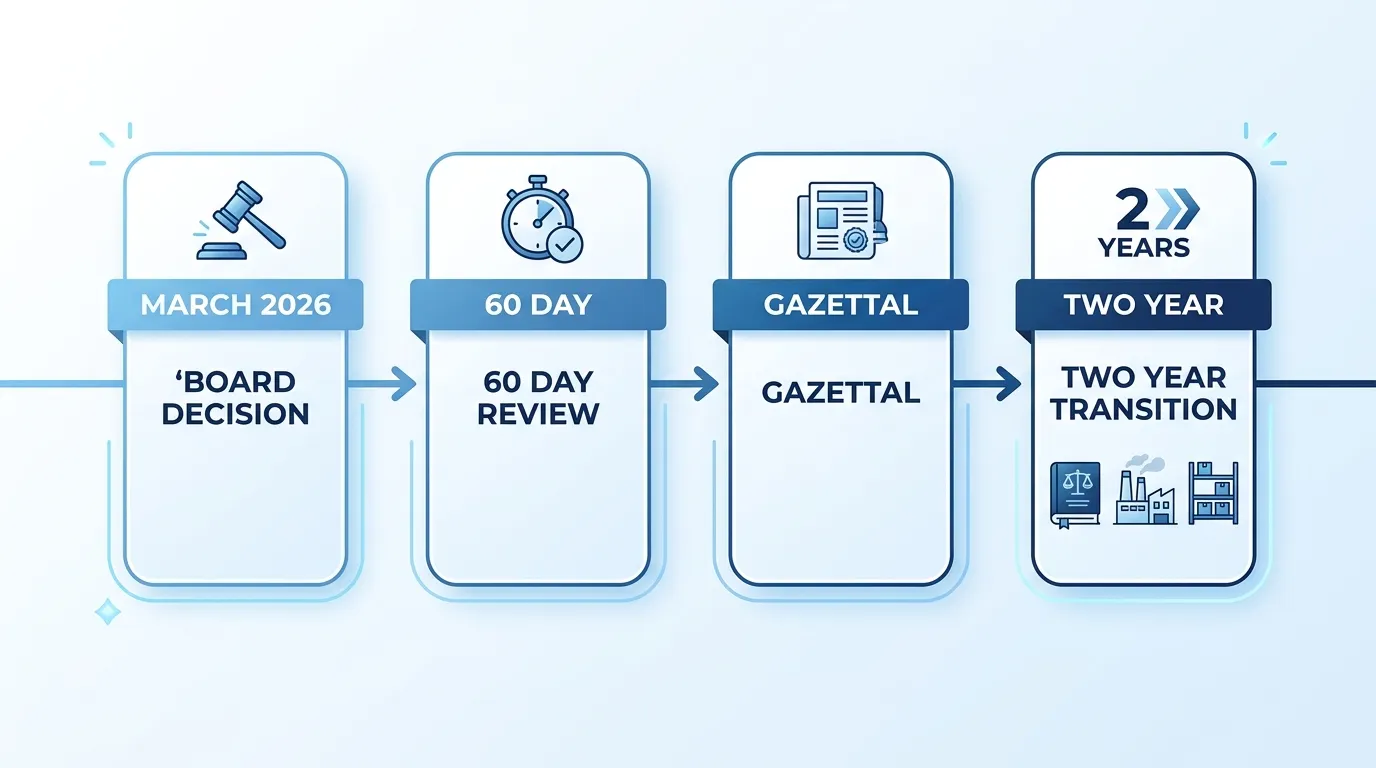
The official P1056 summary groups the amendments into several concrete buckets.
Retail sale of caffeine as a food
The Code will prohibit the retail sale of caffeine as a food. That targets situations where caffeine itself is sold in a form meant for consumers to add to drinks or foods, rather than consuming caffeine only as part of an already regulated product category. The goal is to narrow the channel for dangerously concentrated "raw" caffeine in the general food supply.
Adding caffeine to foods for retail sale
FSANZ frames a broad rule: caffeine from all sources will not be permitted as an added ingredient in foods for retail sale unless the Code explicitly allows it elsewhere. In practice, that means companies cannot freely fortify random snacks or novelty foods with caffeine powder or extracts unless a specific standard permits that use. Permission pathways that remain include categories the Code already regulates in detail (discussed below).
Guarana extract
Guarana contains natural caffeine, but extracts can be produced at concentrations that behave like a concentrated additive in the marketplace. P1056 restricts retail sale of guarana extract with high caffeine concentrations, aiming to stop highly concentrated sources from being sold as ordinary food ingredients without appropriate controls.
Packaged coffee beverages with very high caffeine
For packaged coffee beverages that exceed defined "high caffeine" thresholds, new requirements will apply: declare caffeine content per serve in the nutrition information panel, and carry an advisory statement that the product is not suitable for children under 15, or for pregnant or breastfeeding women, reflecting the populations regulators worry about most when doses climb.
This matters because ready-to-drink coffee products have expanded in strength and marketing reach. A shopper comparing two bottles may soon get a standardized quantitative cue rather than relying on brand names alone.
Formulated supplementary sports foods (FSSF)
Sports nutrition categories receive a structured permission rather than a free-for-all. FSANZ will allow caffeine in formulated supplementary sports foods within defined limits, including a maximum one-day quantity of 200 mg from caffeine (counting caffeine from any source in that product context). Industry reporting also highlights new advisory and warning labelling for these products, including an alternative warning wording that explicitly mentions breastfeeding alongside pregnancy, and an advisory that the product contains caffeine. Multipacks will face additional packaging requirements in some configurations.
What stays the same
FSANZ is explicit about continuity. Existing permissions for caffeine in energy drinks and cola-type beverages remain unchanged, subject to their current standards. Ingredients that naturally contain caffeine, such as coffee or tea used as recognizable ingredients, can still be added to foods unless they qualify as an unapproved novel food under the Code, which triggers a separate pre-market safety pathway.
That distinction matters for reading labels: a mocha bar flavored with coffee is a different regulatory story from a bar with isolated caffeine anhydrous added for effect.